Candle wax melts, moves up the wick, turns into vapor near the flame, and burns into mostly carbon dioxide and water vapor.
Candle wax does not simply vanish. Leftover wax in a jar or wax sitting in a warmer can make the process look less obvious. This page explains the normal burn path first. Then it shows the common reasons some wax stays behind. For the main guide, see candle making.
Where does candle wax go when it burns?
When a candle burns, melted wax moves up the wick, turns into vapor near the flame, and burns into mostly invisible gases, along with heat and light.
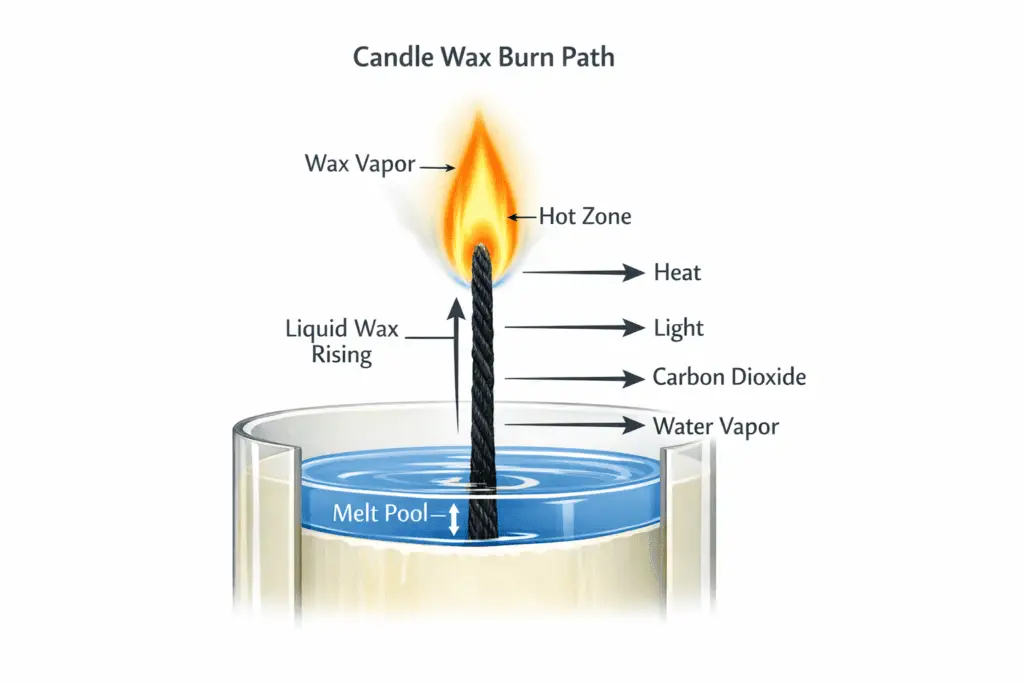
That is why candle wax seems to disappear instead of collecting in the jar forever. The visible melt pool is only the first stage. The part that actually feeds the flame is wax vapor rising from the wick area. Candle wax does not dry up or vanish on its own. It melts first, then burns as fuel once heat turns it into gas. Across different wax types, that same basic path explains where the wax goes when a flame is present.
For a broader look at how formulas behave, see candle wax types.
Process at a glance
- Solid wax softens and forms a melt pool.
- The wick pulls liquid wax upward.
- Heat near the flame turns that liquid into vapor.
- The vapor burns, so less solid wax remains over time.
How the wick turns melted wax into fuel
The wick is a delivery path, not the main fuel.
Heat from the flame melts nearby wax, and the wick draws that liquid upward through its fibers. Near the hottest part of the flame, the wax changes into vapor, and that vapor keeps the candle burning. Melting is only the first step. What matters most is that the wax reaches the hot zone and becomes gas.
A weak or struggling flame usually points to poor wax feed rather than some mystery about disappearing wax. When the wick is too long, too short, clogged, or poorly matched to the candle, melted wax may not reach the flame consistently. In normal combustion, though, the wax is consumed as vapor and burned into mostly carbon dioxide and water vapor.
Burning wax vs melted-only wax
Burning wax is consumed because wax vapor combusts in a flame. Melted-only wax stays wax and simply cools, gets removed, or is reused.
The easiest way to understand the difference is to ask whether a flame is involved. In a candle, melted wax rises through the wick and is consumed after it becomes vapor. In a warmer or tray, the wax may liquefy and release fragrance, but it does not go through the same combustion step.

Quick comparison
- Burning candle: wax melts, rises, vapor burns, and the wax amount drops over time.
- Melted-only wax: wax changes from solid to liquid, then becomes solid again when cooled.
- Warmer wax after scent fade: the smell weakens first, while the wax itself often remains.
This is why wax melts behave differently from candles. A warmer can make wax look active because it is hot and fragrant, but it is not making the wax disappear the way a flame does. Melting tells you when wax becomes liquid, not when it is actually being consumed.
Why wax melts and warmers leave wax behind
Wax melts and warmers usually leave wax behind because the wax is heated enough to release fragrance, but not burned in a flame. Once the scent fades, the wax often remains until you remove it, reuse it, or throw it away.
Why wax sometimes stays in the jar
If wax stays in the jar, the candle usually did not burn under conditions that allowed enough melted wax to reach the flame.
That does not change the normal pathway of melt, wick feed, vapor, and combustion. Leftover wax usually means part of the wax never completed that full trip to the flame, or the candle reached a normal stopping point with some wax still at the bottom. The most common causes are short burn sessions, drafts, poor wick performance, and tunneling.
| Common pattern | Likely cause | What to try next |
| Wax ring on the sides | Short burn sessions | Let the melt pool widen more evenly on the next burn |
| Deep center hole | Undersized wick or repeated short burns | Check burn habits first, then correct wick issues if the pattern continues |
| Uneven burn with extra residue | Drafts or unstable flame | Move the candle away from vents, fans, and strong airflow |
| Small amount left at the end | Normal end-of-life design in some containers | Follow the maker’s stopping guidance rather than forcing a full burn |
Short burn sessions matter because edge wax can stay solid while only the center area keeps feeding the flame. Drafts can pull heat to one side and make a candle burn unevenly, which leaves more wax behind than expected. Wick problems add another layer, because a weak wick cannot move enough liquid wax upward to keep consumption steady.
A practical way to judge what happened is to separate avoidable leftovers from normal leftovers. Avoidable leftovers show up as sidewall wax, tunneling, or uneven patches after poor burn conditions. Normal leftovers are the small amount that may remain when a container candle is simply finished and should not be burned further.
For practical next steps after a candle is finished, see what to do with leftover candle wax.
Do different candle wax types burn differently?
Different wax types can change melt speed, firmness, and visible residue, but they do not change the main answer to this page. In a burning candle, the wax still melts, feeds the wick, turns into vapor, and burns. The bigger differences between formulas belong in a broader wax-comparison discussion, not in the core explanation of where the wax goes.
Quick answers about where candle wax goes
Does candle wax burn or just melt?
Candle wax does both. It melts first, but the part that actually burns is wax vapor near the flame.
Why is there wax left in the jar?
Wax can stay in the jar when the candle tunnels, the flame burns unevenly, the wick underperforms, or the candle reaches its normal stopping point with a small amount left.
Does warmer wax disappear?
Usually no. In a warmer, the wax melts and releases fragrance, but it is not consumed by flame the way candle wax is.
Final takeaway
Candle wax does not disappear by magic. In a burning candle, it melts, rises through the wick, turns into vapor, and burns. In a warmer, it melts and releases scent, but usually stays behind because there is no flame to consume it.
When wax remains in a jar, the reason is usually burn conditions, wick performance, or the candle reaching its normal end point. And while different wax types can influence how a candle behaves, the full system, including wax, wick, additives, container, and airflow, determines how the candle actually burns.
