Stearic acid is a candle additive that hardens wax, helps molded candles release more cleanly, makes wax look more opaque, and can cause tradeoffs when overused.
Stearic acid is a fatty-acid candle additive used to harden wax, support molded release, and change finish in candle making. The focus here is its practical effects in candles, especially added firmness, cleaner mold release in molded formats, and a more opaque finish. Here, “limits” means formulation boundaries and overuse thresholds, not legal rules, toxicology, or one fixed percentage for every wax. Those limits depend on wax system and candle format, so the useful result can change from one formula to another.
For the wider additive category, use the candle additives guide. For exact percentage questions, use stearic acid dosage by wax type instead of treating this page as a formula chart.
What Stearic Acid Does to Candle Hardness
Stearic acid hardens candle wax by making the body firmer, more rigid, and less easy to dent.
In candle additives in candle making, stearic acid is used when a formula needs more structure, sharper edges, and a candle body that feels stronger in the hand. The main effect here is physical firmness. It is not a blanket promise of longer burn time, better scent throw, or better performance in every wax.
At a practical level, stearic acid changes the wax body so the finished candle holds its shape more strongly. That is why the effect tends to matter most in pillars, molded candles, and other formats where body strength and edge definition are easy to see. A soft wax can still make a working candle, but it may mark more easily, lose edge detail, or feel less solid after unmolding.
| Where the change shows up | Lower stearic effect | Higher stearic effect |
| Molded edges | Softer, rounder edges | Sharper, firmer edges |
| Pillar body | Easier to dent or nick | Holds shape more strongly |
| Surface feel | More yielding | More rigid |
| Handling before use | Picks up marks more easily | Feels more resistant to pressure |
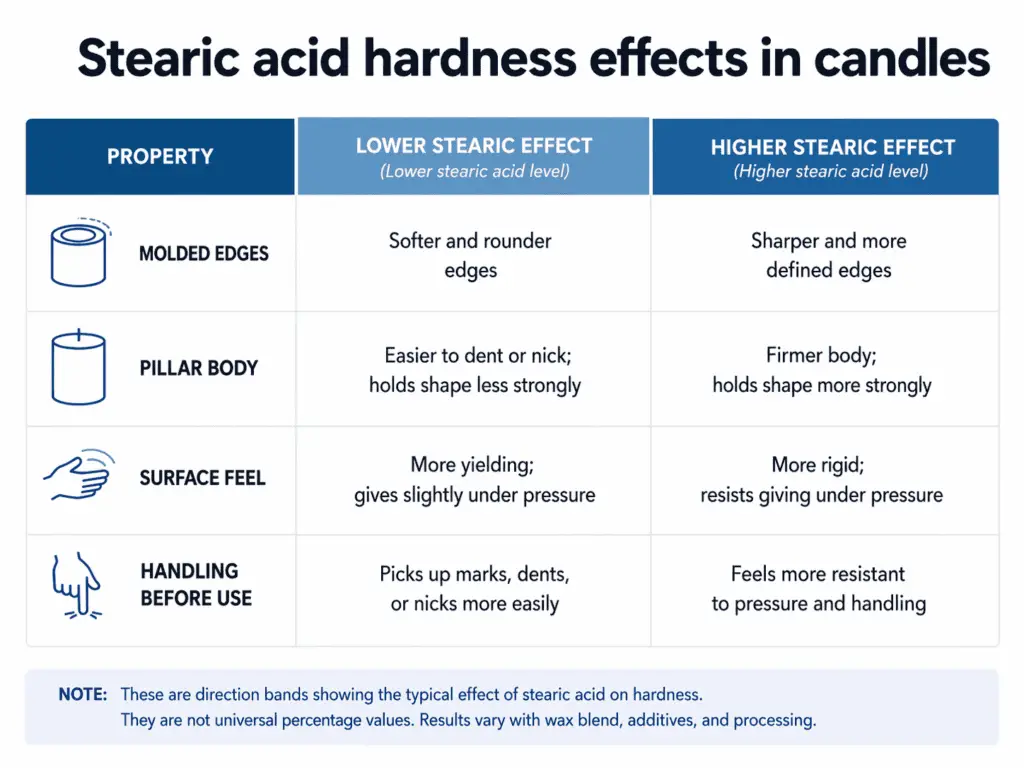
These are direction bands, not a universal percentage chart. They show what the change usually looks like in relative terms without pretending that every wax base responds in the same way.
A common point of confusion is whether stearic acid makes wax only denser or truly harder. In practice, makers use it for the visible and touch-based result: the candle body feels firmer, edges stay cleaner, and the structure resists light damage better. That is why “harder” is the useful word here, even though the exact material change depends on the wax system underneath.
Does this matter more in pillars than in containers? Usually, yes. A container candle is already supported by the jar, so body strength is often less central to the result. A pillar or molded candle has to stand, release, and keep its own shape, so extra rigidity is easier to notice and often more useful. That is a visibility difference, not a rule that one format always needs stearic acid more than another.
Does harder always mean better? No. A harder candle can look cleaner and handle better, but more hardness is not always a better result. Once the wax becomes too rigid for the formula, the gain in firmness can turn into a loss in flexibility, smoother finish, or workable handling.
That is where practical limits of stearic acid in candles start to matter. Hardness is only one effect, and pushing for more of it can create new problems. When the real question is additive choice rather than stearic acid’s effect, stearic acid vs other candle additives belongs on a separate comparison page.
Hardness explains why makers add stearic acid in the first place, but structure matters in another way too: it can help molded candles release more cleanly.
How Stearic Acid Helps Mold Release in Molded Candles
In molded candles, stearic acid can help wax release more cleanly by supporting slight contraction and firmer separation from the mold.
Within candle additives in candle making, this benefit belongs to molded candles and many pillar formulas, not to container candles as a general claim. Stearic acid can help the wax pull away from the mold more neatly as it sets, but mold shape, wax family, pour temperature, cooling pattern, and surface condition still affect the result.
The useful distinction here is release versus contraction. They are connected, but they are not the same thing. Contraction is part of how the candle sets and pulls away. Mold release is the practical result the maker cares about: easier unmolding, cleaner edges, and less sticking when the candle comes out.
That means stearic acid can support release without being the only reason a candle releases well. A clean release can come from the combined effect of the formula, the mold, and the process. In the same way, a sticking problem does not automatically mean the formula needs more stearic acid.
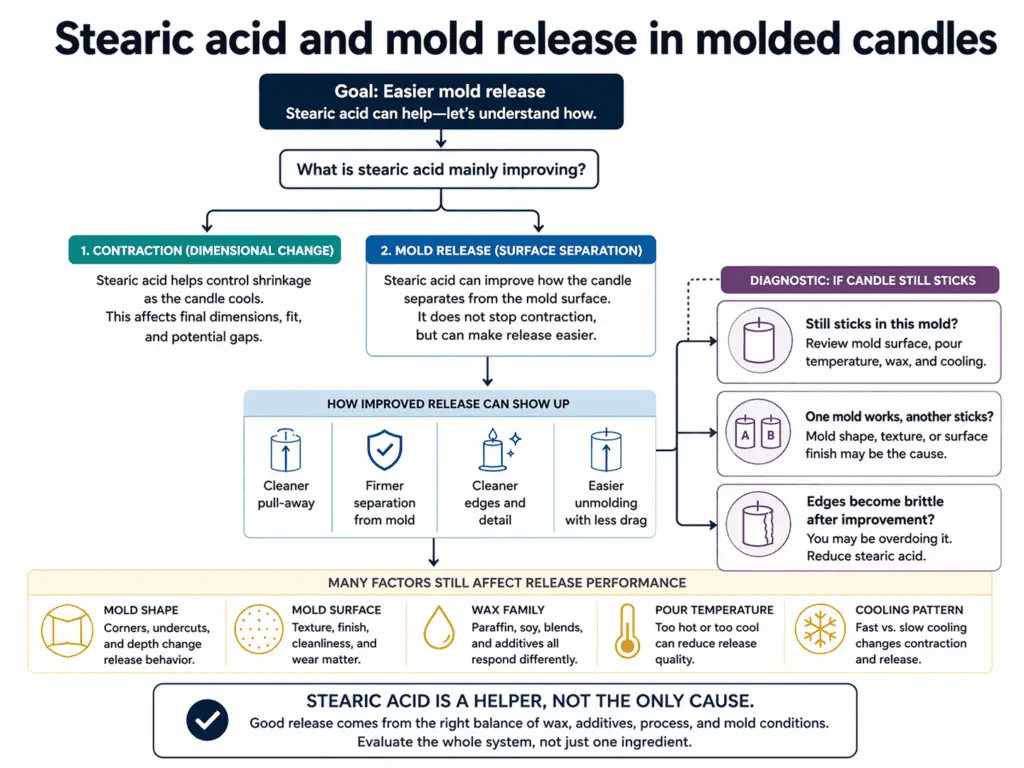
A simple split helps keep the diagnosis on track:
| Problem seen at unmolding | What stearic acid might be helping with | What it does not prove |
| Candle pulls away more cleanly | Firmer structure and cleaner separation | That every mold will behave the same way |
| Edges hold shape better on release | More body strength in the wax | That sink marks or lines are solved |
| Candle still sticks in the mold | Stearic may be too low for this setup | That stearic is the only cause |
| One mold works, another sticks | Formula may be part of it | That mold shape and surface do not matter |
Do all molds benefit equally? No. A simple shape and a difficult shape can react very differently, even with the same wax. A smooth mold surface and a rougher one can behave differently too. That is why stearic acid should be understood as a helper in molded release, not as a universal answer that overrides every mold variable.
Is every sticking problem a stearic problem? No again. Sticking can come from the mold itself, from a poor match between the wax and the format, or from process conditions that keep the candle from setting in a clean way. Stearic acid can improve the odds of easier release, but it does not replace good mold fit or stable cooling.
A short failure check keeps the scope tight:
- If release improves a little after an additive change, stearic acid may be part of the answer.
- If release does not improve at all, the issue may sit more with mold condition or process.
- If release improves but the candle starts showing brittle edges, the formula may have moved too far toward over-hardening.
- If sticking keeps happening after additive changes, the full answer belongs in molded candle troubleshooting rather than in a general note page.
When the issue moves beyond the additive itself and into process problems, molded candle troubleshooting or pillar candle technique should carry the full diagnosis instead of this page.
Stearic acid can help release, but it also changes the way the wax looks after it sets. That visual change is the next part of the story.
What Stearic Acid Does to Opacity and Finish
Stearic acid can make candle wax look more opaque, less translucent, and more solid in finish.
In candle additives in candle making, this is a finish effect caused by the additive changing the look of the set wax. It is not the same thing as adding color. A candle can look more opaque or whiter in finish without that effect acting like a dye.
That distinction matters because makers often use similar words for different results. “Opaque,” “white,” and “solid-looking” may overlap in casual talk, but they do not all mean the same thing in formulation. Stearic acid changes finish and appearance. It does not replace a color system.

| Finish term | What it means here | What it does not mean |
| Opacity | Less light passes through the wax | A dye effect by itself |
| Whitening | The wax can look lighter or more solid | A full white-color strategy |
| Reduced translucency | The candle looks less see-through | A pigment system |
Opacity and whitening are close, but not identical. A candle can become less translucent and look more solid without turning truly white. In the same way, a wax can look slightly lighter or chalkier without becoming a colored candle.
This is why the first question to answer is additive effect versus dye effect. Stearic acid changes the finish of the wax body. Dye changes the color. Those can interact in real formulas, but they are still different functions. When someone wants exact color control, the better route is a candle dye and color additive guide, not a stearic-acid note page.
The second question is whether more opacity is always better. Not necessarily. A more solid-looking candle can look cleaner, especially in molded formats where surface appearance matters. But the same finish shift can become a downside when the surface starts to look dull, overly flat, or chalky.
That is where practical limits of stearic acid in candles show up again. The useful visual change is a more solid finish. The overuse version is a finish that no longer looks clean or balanced for the wax system.
Wax type matters here too. Some waxes show a small finish change. Others show a stronger one. Paraffin and some blends often make the effect easier to see, while other systems may respond differently in strength or in the way the finish appears. That does not mean one group is always right for stearic acid. It means the finish result has to be judged inside the actual wax system, not guessed from a generic claim.
A clean way to think about it is this:
- stearic acid can make wax look less translucent
- that can make the candle look more solid
- that is useful up to the point where the finish starts looking chalky or overworked
So stearic acid affects both how the candle feels and how it looks. Once the visual gain turns into a finish problem, the conversation shifts from benefit to limit.
Practical Limits of Stearic Acid in Candles
Here, limits means the practical point where stearic acid stops helping and starts creating tradeoffs in hardness, finish, or workability.
To keep what stearic acid does in candles clear, this section uses “limits” in the formulation sense only. It does not mean legal rules, toxicology thresholds, or one fixed percentage that works in every wax. It means the usable boundary inside a candle formula, where the additive still helps more than it harms.
That boundary is not best understood as one magic number. It is better understood as a range of behavior. At one end, stearic acid gives a useful gain in firmness, release, or opacity. In the middle, it may still work but with rising tradeoffs. At the far end, the candle starts showing that the additive level no longer fits the wax system or the format.
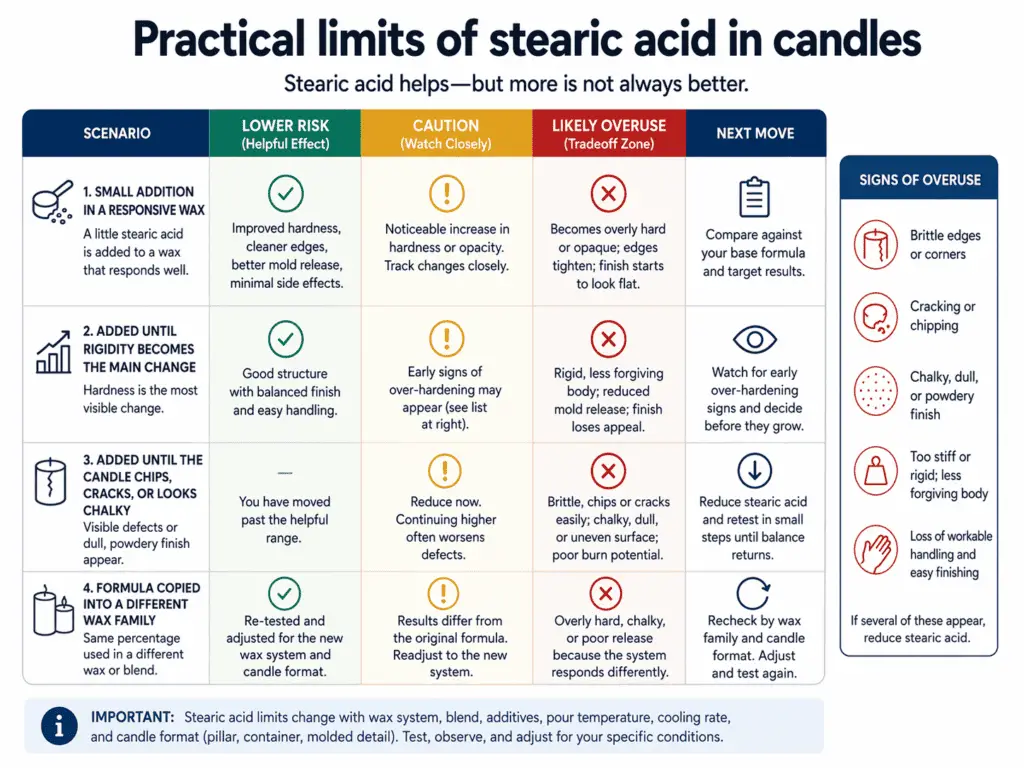
| Situation | Likely result | Risk band | Next move |
| Small addition in a wax that responds well | Firmer body, cleaner release, more opacity | Lower risk | Compare against the base formula |
| Added until rigidity becomes the main visible change | Sharper structure but less flexibility | Caution | Watch for early over-hardening signs |
| Added until the candle chips, cracks, or looks chalky | Useful effect has become a tradeoff | Likely overuse | Reduce and retest |
| One formula copied into a different wax family | Results shift faster than expected | Caution to likely overuse | Recheck by wax and format |
So what counts as too much? In this page’s scope, “too much” means the point where the next visible change is no longer a gain. If a little more stearic acid gives you sharper edges but also creates brittle corners, you have already crossed from benefit into tradeoff. If it gives you a more solid finish but also pushes the wax toward a chalky look, the same rule applies.
That is why risk-band thinking works better here than a single number. This page can tell you how to recognize the boundary. It should not pretend that one exact percentage belongs to every wax family, every blend, and every candle format.
A practical check looks like this:
- Start with the effect you actually need: more firmness, easier release, or a less translucent finish.
- Compare the result against the base formula instead of chasing the strongest possible change.
- Stop when the next gain starts bringing stiffness, chalkiness, cracking, or loss of workable handling.
- Treat that point as the practical boundary for this wax and format.
- Move exact percentage questions to stearic acid dosage by wax type rather than forcing one answer onto every system.
That same boundary logic answers another common confusion: “limits” here are not safety or compliance limits. Questions about exposure, handling, or label-level safety belong under stearic acid safety and SDS guidance. Questions about recipe behavior belong here.
When Hardness Turns Into Brittleness
Too much stearic acid can push a candle past the practical limits of stearic acid in candles and into a brittle, crack-prone state.
This is the point where useful hardness turns into a tradeoff. A candle may feel strong at first, but the wax body has become too rigid for the formula or format.
Common signs that point toward over-hardening:
- edges chip instead of flexing slightly
- corners break during unmolding
- the candle feels snap-prone rather than simply firm
- surface appearance starts looking dry or chalky
- the structure feels overbuilt for the wax and candle type
When cracking, chipping, or release problems need full diagnosis, that work belongs in candle defect troubleshooting rather than on this page.
Why Limits Change by Wax System
Practical limits vary because different wax systems do not respond to stearic acid in the same way.
Paraffin, blends, and soy-heavy systems do not all respond to stearic acid with the same strength or at the same point. Candle format changes the result too, so a usable level in one wax-and-format setup should not be treated as universal.
When the question becomes how much stearic acid to use in a specific wax family or candle format, that belongs on stearic acid dosage by wax type rather than inside this overview page.
