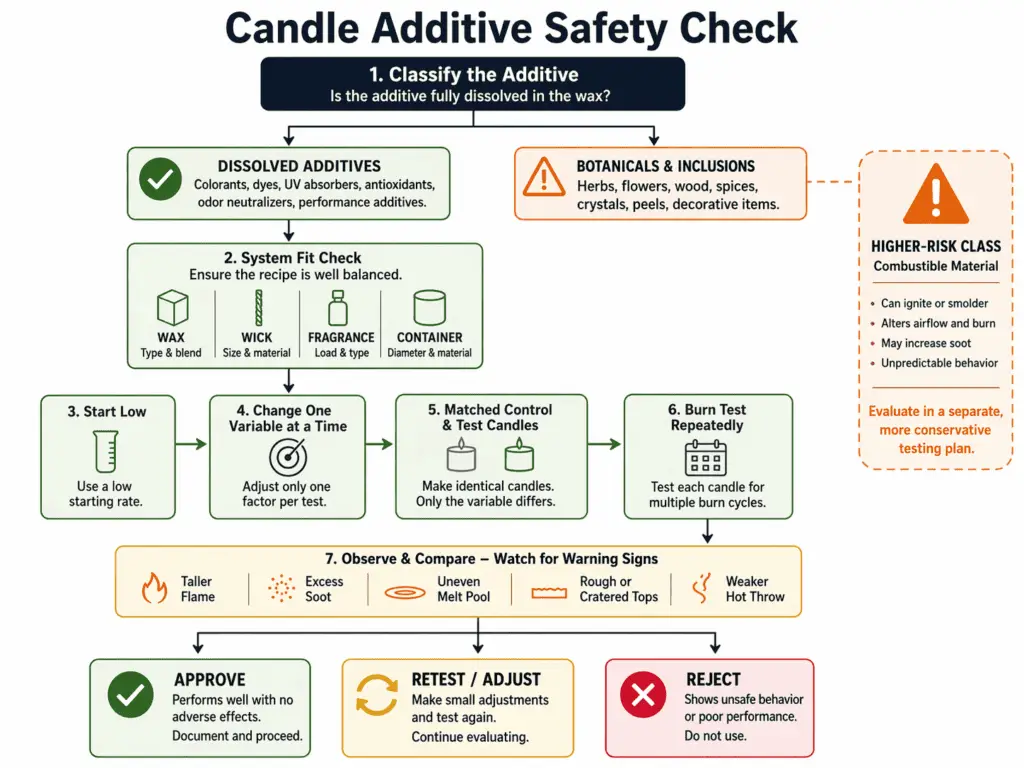
Common dissolved candle additives are safe only when they fit the wax, wick, fragrance, and container system and pass repeatable burn testing after recipe changes, while botanicals belong to a separate higher-risk class.
Candle additives such as Vybar, stearic acid, and UV inhibitors are materials added to wax to change burn behavior, fragrance retention, hardness, appearance, or fade resistance. Here, safe means matched to the wax, wick, fragrance, and container system, used at a cautious starting rate, and confirmed by burn testing. It does not mean skin-safe, food-safe, harmless in every amount, or legally cleared for every market. Botanicals sit in a different risk class because they can act as combustible material instead of dissolving into the wax. For broader context, see candle additives.
This page covers burn-system safety and formula stability in candle making. It does not cover toxicology, indoor-air claims, legal compliance, or exact dosage tables.
What “Safe” Means for Candle Additives
Candle additives can be safe in candles when they fit the full candle system, stay within cautious starting ranges, and are confirmed by burn testing.
Candle additives are candle-making materials added to wax to change scent hold, hardness, appearance, or color stability. A broader overview of purposes sits under Candle Additives for Scent, Hardness, and Stability, but the safety question is narrower. On this page, safe means the additive suits the wax system, the amount stays conservative, and the finished candle burns in a stable and repeatable way. It does not mean that every additive is harmless, that natural materials are low risk, or that a candle is ready for sale just because one test burn looked fine.
The safest way to think about additives is to treat them as small recipe controls, not rescue tools. A good additive choice can improve a candle that is already close to working. A bad additive choice can hide the real problem for one pour and then create new trouble in the next test. That is why safety depends on the whole recipe, not on the ingredient name alone.
Botanicals are not safe in the same way as dissolved additives
Botanicals are not safe in the same way as dissolved additives because they can act as combustible material instead of dissolving into the wax. Dried flowers, herbs, seeds, spices, crystals, shells, and similar items can sit near the flame, move as the wax softens, or leave char behind. That is why Are Botanicals Safe in Candles? belongs in a separate discussion, even though makers often mention botanicals in the same breath as additives.
The word natural causes a lot of confusion here. A dried flower may look milder than a manufactured additive, but the real question is how it behaves near heat and flame. If it can ignite, curl into the melt pool, shift toward the wick, or leave burning debris, it has a different kind of risk than an additive that dissolves into the wax.
What safe use usually looks like
Safe additive use usually starts with a narrow goal. You add something because you want a modest change in hardness, opacity, scent hold, or color stability. You begin at a low supplier-start range, change one variable at a time, and compare matched test candles. You then judge the result by burn behavior, surface quality, and repeatability instead of by hope.
A practical rule is simple: the safest amount is the lowest amount that does the job without creating fresh defects. More additive is not better by default. Once the amount starts pushing the flame higher, changing the melt pool too much, dulling the throw, roughening the surface, or making the wax harder than needed, the rate is no longer helping.
Signs an additive rate is too high
Too much additive is any amount that moves the candle away from stable performance.
| What you are checking | Better sign | Warning sign |
| Flame behavior | Steady flame with no new soot | Taller flame, flicker, soot, or a stressed burn |
| Melt pool | Even and repeatable | Strange pooling, uneven consumption, or a sudden behavior shift |
| Surface finish | Small visual improvement | Rough tops, drag marks, cloudy spots, or a surface change with no payoff |
| Scent throw | Balanced result after cure and testing | Weaker hot throw after adding more |
| Recipe control | One change tested at a time | Several changes at once, so the cause stays unclear |
Why system fit matters more than ingredient labels
A candle is a system made of wax, wick, fragrance, dye, vessel, room conditions, and formula choices. An additive that behaves well in one system can create problems in another. A paraffin blend may respond very differently from a soy-heavy blend. A wick that was already near its limit may react badly to a harder or denser wax. A fragrance that was already heavy can change the result again.
That is why the next question after additive choice is never “Is this ingredient safe by itself?” The better question is “Does this ingredient improve this exact recipe without creating a worse burn?” The answer only comes from matched testing.
When you need fresh testing
Any meaningful recipe change resets the old result. That includes a new additive, a different additive level, a wax swap, a fragrance-load change, a wick change, or a vessel change. The route back to a reliable answer is How to Burn Test a Candle After Recipe Changes. One clean burn is not enough. You want repeated burns of the same version so the pattern is clear.
A minimum check is this: change one variable at a time, label the control and test versions, and burn them under matched conditions. Watch the flame, soot, melt pool, and repeatability, repeat the burns, and reject any version that creates new instability.
It also helps to separate recipe safety from legal or labeling work. Sale rules, fragrance documents, and market duties belong under CLP vs SDS vs IFRA for Candle Makers, not inside the core additive answer. Keeping those boundaries clear makes this page easier to trust because it stays on candle-additive safety instead of drifting into a second topic.
How Safe Is Vybar When Used in Candle Wax?
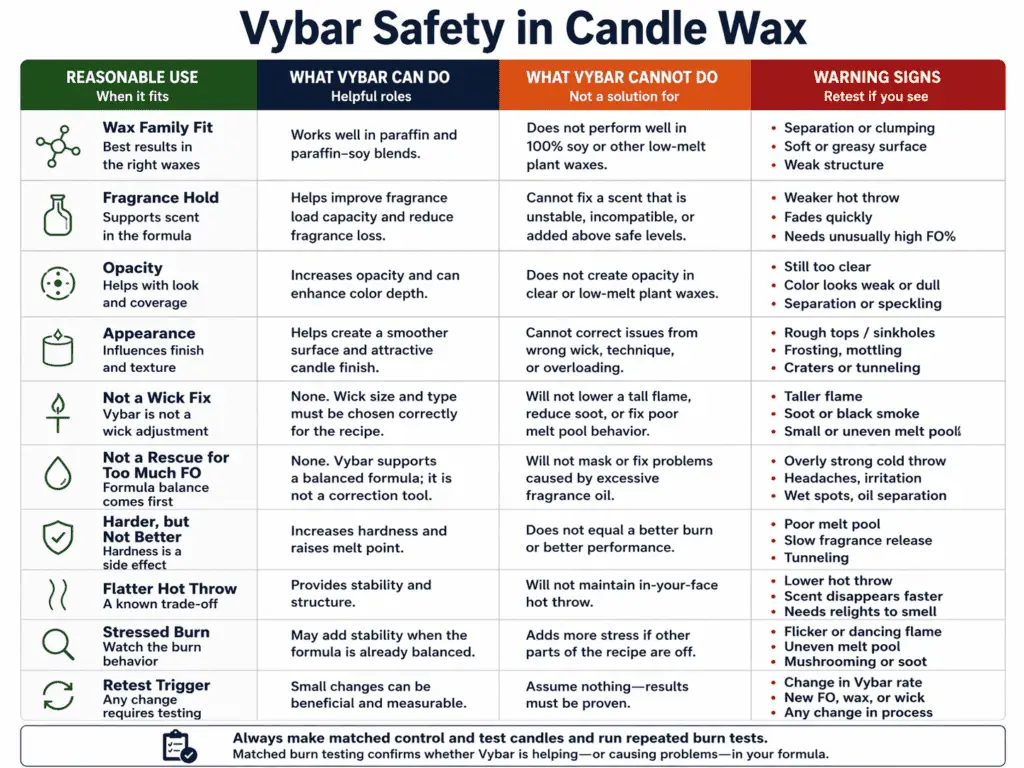
Vybar can be appropriate in candle wax when the grade suits the wax family, the amount stays cautious, and the final recipe passes burn testing.
Vybar is a candle additive used in some candle formulas to improve fragrance hold, opacity, and general wax behavior. Safe use starts with the right match between the Vybar grade and the wax family. It continues with a modest starting amount and matched test pours. It does not mean Vybar belongs in every candle or that it can fix a recipe that is off-balance for other reasons.
When Vybar is usually a reasonable choice
Vybar is usually considered for paraffin or certain blended wax systems where a maker wants modest help with scent hold or appearance. The key point is fit. Wax Compatibility for Candle Additives matters because one formula can welcome a small amount of Vybar while another can react poorly or gain little from it.
What Vybar can and cannot do
Vybar can sometimes tighten up a formula. It may help with opacity, appearance, or how a fragrance behaves in certain systems. What it cannot do is rescue a poor wick decision, correct a badly chosen wax, or erase the effects of too much fragrance oil.
Vybar compared with stearic acid
The phrase Vybar vs Stearic for Candles matters because both ingredients can change candle behavior, but they are not the same tool. Vybar is often used to affect fragrance hold and appearance in certain systems. Stearic acid is more often used to harden or opacify wax. They may overlap in effect at the surface level, but the reason for using them should still be specific. Swapping one for the other without a clear job can push the formula in the wrong direction.
Clear warning signs with Vybar
Vybar is becoming risky when each increase makes the wax harder but not better, when the hot throw seems flatter, or when the burn looks more stressed after the change. It is also a warning sign when the maker cannot explain why Vybar was added beyond “the candle was not working.”
That is the moment to stop adding and retest the recipe itself. The route back is How to Burn Test a Candle After Recipe Changes with matched samples and one variable changed at a time. For deeper Vybar-specific guidance, see Vybar in candles instead of extending this safety note.
What Makes Stearic Acid Safe or Risky in Candles?
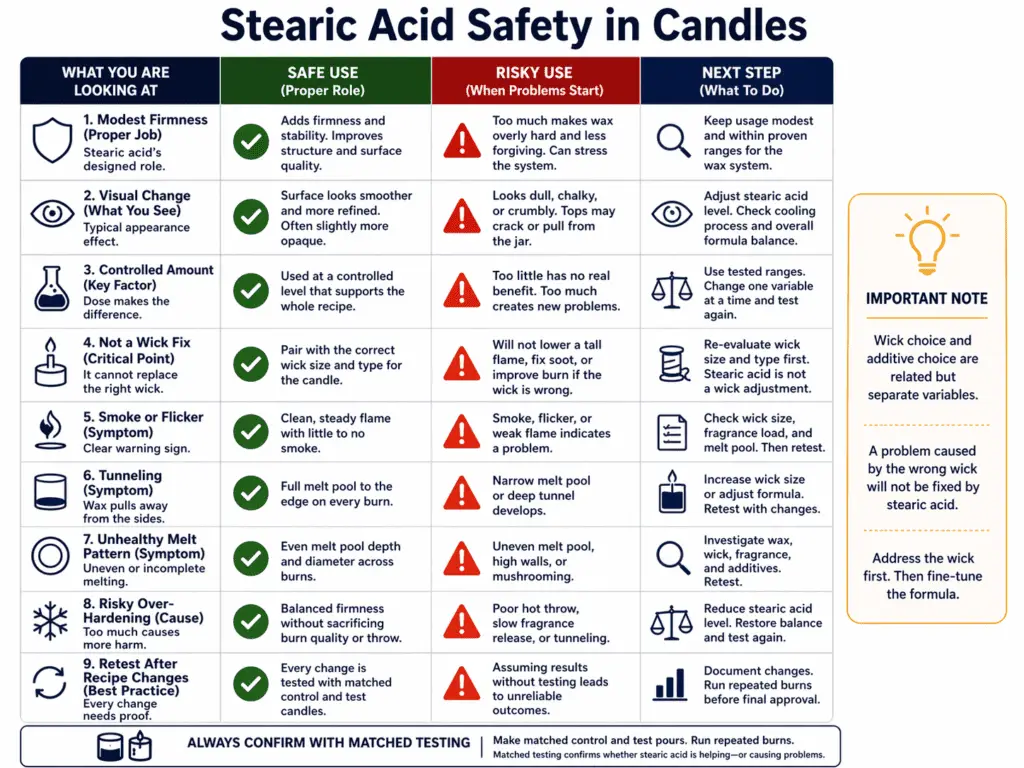
Stearic acid can be appropriate in candles when it has a clear formula job, starts low, and is retested after the recipe changes.
Stearic acid is a candle additive used to harden wax and change how a candle looks or behaves. In candle terms, safe does not mean skin-safe, food-safe, or harmless in every setting. It means the additive fits the wax system, the amount stays controlled, and the candle still burns in a stable way after the change. That distinction matters because stearic acid appears in other product types, but this page is only about candle use.
When stearic acid may help
Stearic acid may help when a formula needs modest added firmness or a visual change that fits the chosen wax family. It works best when the maker knows the exact job it is supposed to do. “I want a slightly firmer candle” is a real goal. “My candle is bad, so I added stearic” is not a reliable reason.
Stearic acid is not a wick fix
This is where How Wick Choice Changes Candle Safety becomes important. A wick controls how heat enters the system. Stearic acid changes the wax system. Those are related, but they are not the same variable. If a candle smokes, flickers, tunnels, or fails to reach a healthy melt pattern, adding stearic acid can move the behavior without telling you whether the wick was ever correct.
Stearic acid compared with Vybar
The phrase Vybar vs Stearic for Candles matters here for the same reason it mattered in the Vybar section: these are not direct substitutes. Stearic acid is often chosen for hardness and opacity. Vybar is often chosen for other changes tied to certain wax systems. A maker should choose the additive based on the problem being solved, not based on the fact that both ingredients show up in candle recipes.
What makes stearic acid risky
Stearic acid becomes risky when the formula keeps getting harder while the actual candle performance gets less clear. It is also risky when the additive is added because of non-candle uses or because it seems familiar from soap or skincare. Familiarity outside candle making says nothing about burn behavior inside a candle.
The right response after any change is How to Burn Test a Candle After Recipe Changes with matched pours and repeated burns. For deeper stearic-specific guidance, see Stearic acid in candles instead of extending this safety note.
Why and When Is UV Inhibitor Safe to Use in Candles?
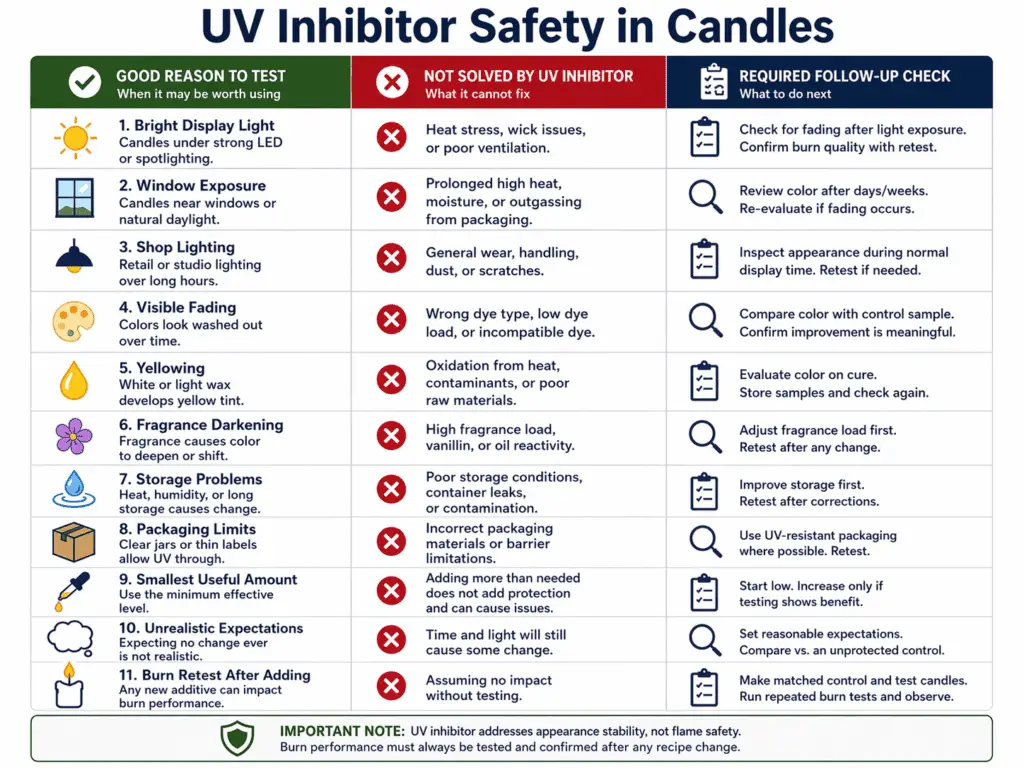
UV inhibitor can be appropriate in candles when it is used for real light-exposure problems, started at a cautious supplier range, and kept within realistic limits.
UV inhibitor is a candle additive used to help reduce visible fading or yellowing in some candles. In candle terms, safe means candle-only use, a controlled starting amount, and honest expectations about what the additive can do. It does not mean total protection from color change, and it does not mean the candle is safer around flame. UV inhibitor is about appearance stability, not fire control.
When UV inhibitor makes sense
UV inhibitor makes the most sense when a line spends time in bright display light, near windows, or under shop lighting that repeatedly causes visible fading. It may also be worth testing when certain colors or fragrance combinations show a pattern of early change.
What UV inhibitor cannot do
UV inhibitor cannot promise that a candle will never discolor. It cannot correct poor storage, fix a fragrance that darkens over time, or replace sensible packaging choices. That is where Why Candles Fade or Yellow and Candle Storage and Shelf-Life come into the decision. A maker who ignores storage and display conditions may blame the additive when the real issue sits elsewhere.
This is also why more UV inhibitor is not automatically better. A higher amount can crowd the recipe without solving the cause of the color change. The useful amount is the smallest one that produces a visible gain in a fair test.
Safe expectations for UV inhibitor
A safe expectation is modest: slower visible change in a formula that has a real light-exposure problem. An unsafe expectation is total color protection in every condition. That second expectation pushes makers toward overuse and disappointment.
When to retest
Any addition of UV inhibitor changes the recipe, so the next step is How to Burn Test a Candle After Recipe Changes. Even though the additive is being used for appearance stability, the candle still has to burn well after the change. The right formula is the one that holds up better in light without causing new burn problems.
A careful maker tests matched candles, keeps the exposure conditions fair, watches for color change over time, and then confirms that burn behavior stayed stable. For deeper UV-inhibitor guidance, see UV inhibitor in candles instead of extending this safety note.
